Dublin, April 20, 2026 (GLOBE NEWSWIRE) — The “Biologics Contract Research Organization Market: Industry Trends and Global Forecasts – Distribution by Type of Biologic, Scale of Operation, Therapeutic Area, and Geography” report has been added to ResearchAndMarkets.com’s offering.
The global biologics contract research organization market valued at USD 36 billion in the current year and is anticipated to reach USD 126 billion by 2035, growing at a lucrative CAGR of 13% during the forecast period.
Biologics are among the fastest expanding sectors within the pharmaceutical industry. This can be linked to the swift advancement in this area, fueled by the demand for efficient and tailored pharmacological solutions. The increased need for biologics including monoclonal antibodies, gene therapies, and cell therapies is driven by their success in addressing chronic and complex ailments such as cancer, autoimmune diseases, and neurological disorders.
This need is intensified by an expanding biologics pipeline and elevated approval rates. Nonetheless, the development and clinical evaluation of biologics is a very intricate and expensive procedure that requires specialized knowledge and cutting-edge bioprocessing technologies. Consequently, numerous biopharmaceutical firms are opting to delegate their drug discovery and clinical research activities.
At present, biopharmaceutical firms are adopting a business model wherein development processes are contracted to external service providers, while the firm itself oversees the R&D of these biologics. The biologics contract research organization sector is experiencing growth fueled by the rising research on innovative medications and illnesses. R&D of biologics is labor-intensive and pharmaceutical companies need significant technological knowledge and specialized expertise, which increases the overall expense of drugs. Moreover, strict regulatory standards and the approval process can delay the approval of drugs, resulting in a drug supply shortfall in the market.
To simplify these intricate processes and address related challenges, leading pharmaceutical companies are progressively delegating their biologics research and development efforts to specialized contract research organizations (CROs). This strategic outsourcing enables them to utilize external knowledge, speed up drug discovery and clinical trials, and ultimately launch innovative biologics to market more effectively.
In addition, the integration of AI can speed up drug discovery, enhance clinical trials (such as patient recruitment), and boost manufacturing yields. The domain presents numerous opportunities in implementing automation for bioprocessing and decentralized / virtual clinical trials, which lower expenses and improve data quality. As a result, the biologics contract research organization market is expected to grow steadily to meet the increasing R&D needs of biopharmaceutical sponsors.
BIOLOGICS CONTRACT RESEARCH ORGANIZATION MARKET: KEY SEGMENTS
Clinical Segment is Likely to Hold the Highest Share in the Biologics Contract Research Organization Market
In terms of the scale of operation, the global market for biologics contract research organization market is segmented into discovery, preclinical and clinical. Currently, majority share of the share is captured by clinical scale of operation, followed by preclinical and discovery segments.
North America is Anticipated to Capture the Maximum Biologics Contract Research Organization Market Share
In terms of geographical regions, the global market is segmented into North America, Europe and Asia-Pacific. Amongst these, North America is likely to capture majority (50%) of the market share, followed by Europe (32%) and Asia-Pacific (16%).
BIOLOGICS CONTRACT RESEARCH ORGANIZATION MARKET: KEY INSIGHTS
The report delves into the current state of the biologics contract research organization market and identifies potential growth opportunities within industry.
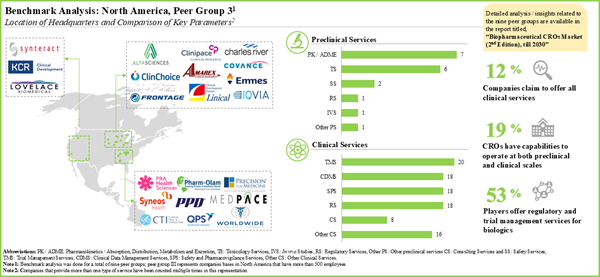
- Presently, 160 players claim to have the necessary capabilities to offer contract research services and clinical trial support for different types of biopharmaceutical products.
- About 50% of all the players offer only clinical services; of these, 12% CROs provide all the services associated with clinical research of biologics.
- The current market landscape is fragmented, featuring a mix of well-established players and specialty service providers, having extensive portfolios to support preclinical and clinical research.
- It is estimated that big pharma players presently outsource close to 45% of their internal R&D operations to CROs.
- More than two-thirds of the contract research service providers are based in North America and Europe; most of these players are small and mid-sized companies.
- In order to gain a competitive edge, companies are actively expanding their existing capabilities in order to further augment their respective offerings and also comply with evolving industry benchmarks.
- The growing interest of stakeholders in this field is also reflected in the increase in partnership activity in the recent past; since 2016, industry players have signed multiple deals with sponsor and / or other CROs.
- Over 8,000 product candidates are currently being evaluated / under development biologic drug developers, presenting opportunities in the biologics contract research organization market.
- Close to 20% of the CROs provide research services for biologics at the clinical and preclinical scales.
- With the rising demand for one-stop shops, industry stakeholders are actively consolidating their capabilities related to biopharmaceutical research, mostly through mergers and acquisitions, and internal expansions.
- The market is expected to grow at a CAGR of ~13% in the coming decade; the opportunity is likely to be well distributed across therapeutic areas, scales of operation, types of biologics, end users and geographical regions.
REASONS TO BUY THIS REPORT
- The report provides a comprehensive market analysis, offering detailed revenue projections of the overall market and its specific sub-segments. This information is valuable to both established market leaders and emerging entrants.
- Stakeholders can leverage the report to gain a deeper understanding of the competitive dynamics within the market. By analyzing the competitive landscape, businesses can make informed decisions to optimize their market positioning and develop effective go-to-market strategies.
- The report offers stakeholders a comprehensive overview of the market, including key drivers, barriers, opportunities, and challenges. This information empowers stakeholders to stay abreast of market trends and make data-driven decisions to capitalize on growth prospects.
KEY QUESTIONS ANSWERED IN THIS REPORT
- How many companies are currently engaged in this market?
- Which are the leading companies in this market?
- What factors are likely to influence the evolution of this market?
- What is the current and future market size?
- What is the CAGR of this market?
- How is the current and future market opportunity likely to be distributed across key market segments?
COMPANY PROFILES
- Biocon
- Company Overview
- Financial Information
- Services Portfolio
- Future Outlook
- Covance
- ICON
- Medpace
- Pharmaron
- PPD
- PRA Health Sciences
- Syneos Health
- Vimta Labs
- WuXi AppTec
BENCHMARK ANALYSIS
- Methodology
- Benchmark Analysis: Peer Groups
- Peer Group I
- Peer Group II
- Peer Group III
- Peer Group IV
- Peer Group V
- Peer Group VI
- Peer Group VII
- Peer Group VIII
PARTNERSHIPS AND COLLABORATIONS
- Partnership Models
- Biopharmaceutical CROs: Recent Partnerships and Collaborations
- Analysis by Year of Partnership
- Analysis by Type of Partnership Model
- Analysis by Year of Partnership and Type of Partnership Model
- Most Active Players: Analysis by Number of Partnerships
- Analysis by Scale of Operation
- Analysis by Geography
- Country-wise Analysis
- Intercontinental and Intracontinental Agreements
MERGERS AND ACQUISITIONS
- Merger and Acquisition Models
- Biopharmaceutical CROs: Mergers and Acquisitions
- Analysis by Year of Acquisition
- Analysis by Type of Collaboration
- Analysis by Geography
- Continent-wise Distribution
- Country-wise Distribution
- Ownership Change Matrix
- Most Active Acquirers: Analysis by Number of Acquisitions
- Distribution by Key Value Drivers
- Analysis by Key Value Drivers
- Analysis by Key Value Drivers and Year of Acquisitions
- Valuation Analysis: Acquisition Deal Multiples
MARKET SCOPE
Type of Biologic
- Vaccines
- Cell Therapy
- Gene Therapy
- Antibodies
- Recombinant Proteins / Peptides
- Others
Scale of Operation
- Clinical Operations
- Preclinical Operations
Therapeutic Area
- Oncological Disorders
- Cardiovascular Disorders
- Inflammatory Disorders
- Neurological Disorders
- Other Therapeutic Areas
Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and North Africa
- Rest of the World
ADDITIONAL BENEFITS
- Complimentary PPT Insights Packs
- Complimentary Excel Data Packs Covering All Analytical Modules
- Up to 15% Complimentary Content Customization
- In-Depth Report Walkthrough with the Research Team
- Complimentary Report Update if the Report is 6+ Months Old
For more information about this report visit https://www.researchandmarkets.com/r/dqn2sf
About ResearchAndMarkets.com
ResearchAndMarkets.com is the world’s leading source for international market research reports and market data. We provide you with the latest data on international and regional markets, key industries, the top companies, new products and the latest trends.
