NEW YORK, March 26, 2026 (GLOBE NEWSWIRE) — As non-invasive musculoskeletal care continues to gain attention in the U.S. the launch of a new shockwave therapy system by SHEYERA comes at a time when shockwave treatment is increasingly being discussed in relation to chronic soft-tissue conditions such as tendinopathy and plantar fasciitis. Public clinical information has described it as a conservative treatment option that may help support pain relief and tissue recovery with minimal downtime. Against this backdrop, buyer expectations are also moving beyond basic output power alone, with growing attention on systems that can deliver greater consistency, usability, and control in real treatment settings.
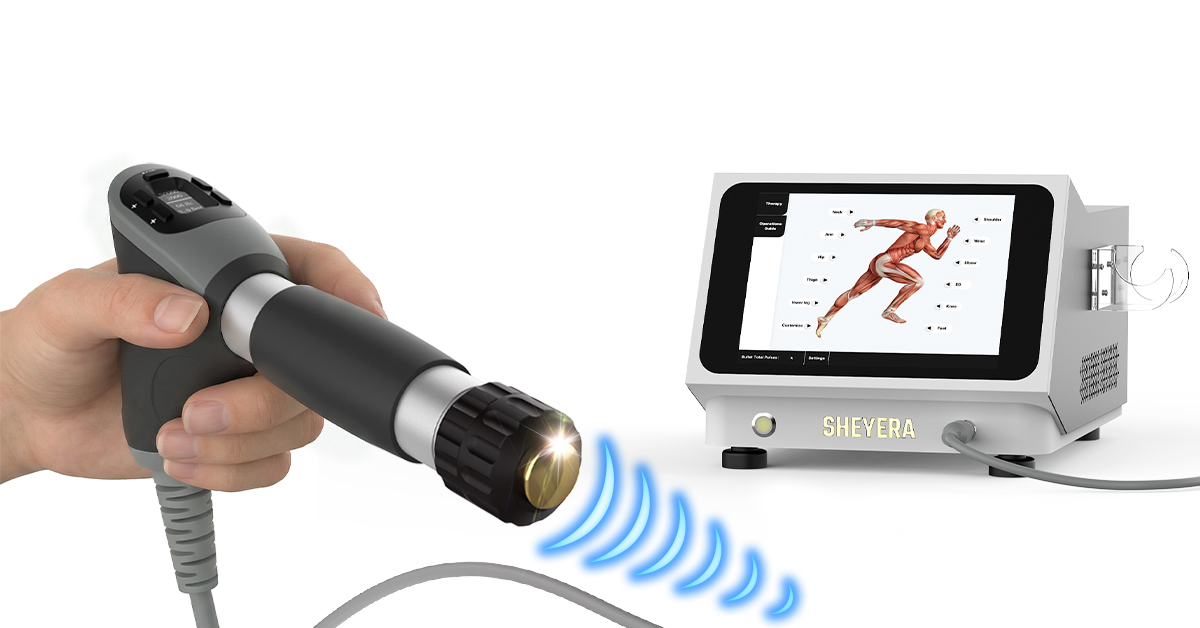
Among newer systems in the category, the SHEYERA MS500 is notable for its workflow-oriented design. It features 47 preset modes, and each preset mode supports one user-saved custom parameter set, a structure that can help clinics and repeat-treatment environments maintain a more standardized process while still preserving individualized settings. For operators, that means less setup uncertainty, smoother repeat visits, and a more polished day-to-day treatment experience.
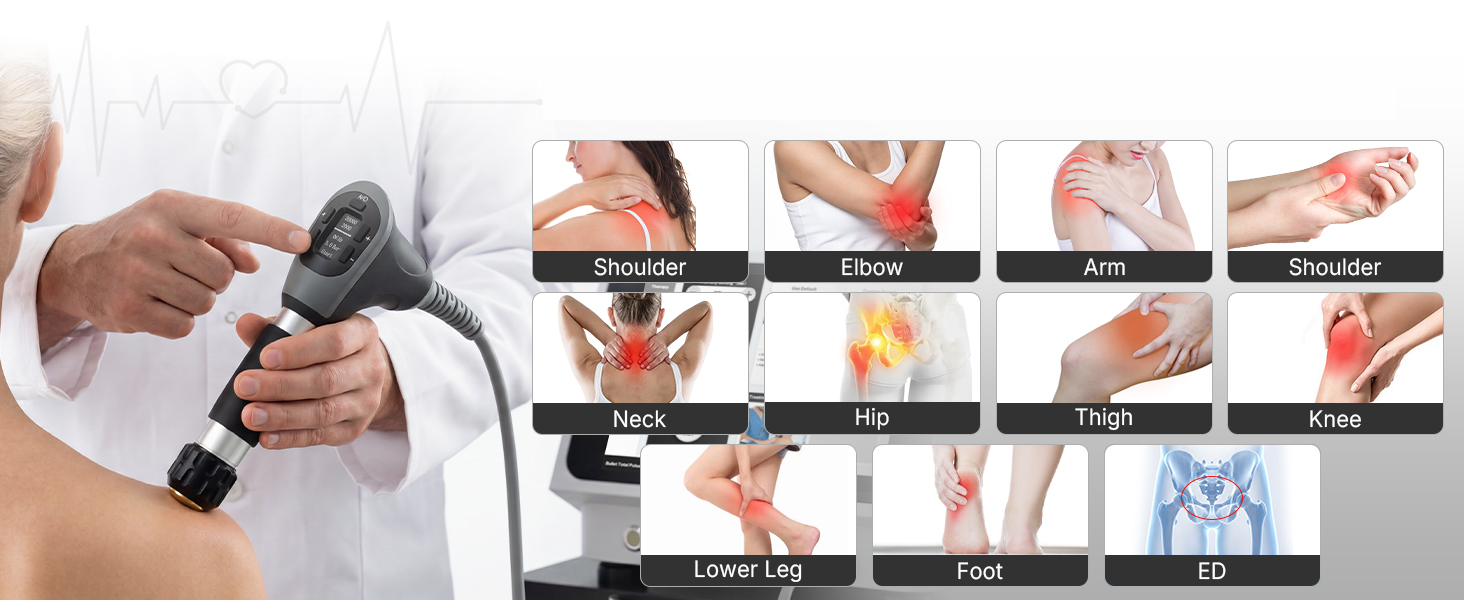
The system is also equipped with 10 applicators designed to support a wider range of treatment scenarios. In addition to standard radial heads, it includes a silicone sensitive applicator for lower-tolerance patients or delicate areas, a joint-specific applicator for improved fit around joint regions, and a patented focused emitter engineered to deliver a more concentrated, higher-density energy pattern when greater precision is needed. Taken together, these features make the SHEYERA a device worth watching for buyers seeking more controlled and adaptable shockwave treatment delivery.

Learn More & Purchase
For more information about the SHEYERA MS500 Shockwave Therapy Device, visit Amazon product page (https://www.amazon.com/dp/B0G4D9KG3M).
Media Contact:
Contact Person: Melody
Email: [email protected]
Photos accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/72622c78-d774-41ce-bf75-7b1763d1e9db
https://www.globenewswire.com/NewsRoom/AttachmentNg/d2e4a003-4beb-4b34-ab49-f4499c1ca91b
https://www.globenewswire.com/NewsRoom/AttachmentNg/a4941b63-60fc-48a8-ad79-fc1f79be5636
https://www.globenewswire.com/NewsRoom/AttachmentNg/6c01c45a-8058-45c0-a5bd-e0b59db4a492

