Short seller’s Characterization of ASCENIV’s Competitive Position Reflects Blatant Misunderstanding of ADMA’s Business and Its Role as Late-line Therapy for Patients Who are Immune Compromised and Therefore, May Not Respond to Vaccines
Highlights that Demand for – and Utilization of – ASCENIV Has Grown Steadily Over Past Two Years and is at Record High
Details that Stocking Levels Maintained by ADMA’s Distributors Align with Industry Standards and Reflect the Long Production Cycle for ASCENIV
RAMSEY, N.J. and BOCA RATON, Fla., March 27, 2026 (GLOBE NEWSWIRE) — ADMA Biologics, Inc. (Nasdaq: ADMA) (“ADMA” or the “Company”), a U.S. based, end-to-end commercial biopharmaceutical company dedicated to manufacturing, marketing and developing specialty biologics, today responded to a report issued on March 24, 2026 by short seller Culper Research (the “Short Report”). Following a detailed review of the Short Report, ADMA wishes to refute key allegations to alleviate confusion in the marketplace concerning the Company’s business practices and operations.
ADMA believes that by providing, on a non-recurring basis, additional details concerning the Company’s immune globulin product portfolio – including inventory days on hand at its distribution partners and direct customers, and end user demand pull-through data – it refutes the key false and misleading allegations contained in the Short Report.
Specifically:
- Demand for ASCENIV is real and growing.
- As shown through data provided directly from ADMA’s distribution partners and direct customers, end-user demand for ASCENIV has increased over the past two-plus years.
- Allegations of channel stuffing illustrate a misunderstanding of the commercial dynamics of the IVIG market.
- Distributors must maintain a level of safety stock to ensure the continuity of care for immune compromised patients who require IG therapy every 21-28 days.
- ADMA’s distributors and direct customers typically stock above contractually required minimum levels to have supply on hand for immediate administration and to mitigate any potential supply chain, manufacturing, testing or regulatory disruptions.
- The Company’s distributors provide inventory and pull through sales data to ADMA on an ongoing basis.
- As shown through data provided directly from ADMA’s distribution partners and direct customers, the graphs entitled “Distributor Inventory Days of Coverage Above Safety Stock” illustrates, as of January 5, 2026 and March 22, 2026, an average of 84 and 48 days on hand, respectively, for ASCENIV, and an average of 87 and 51 days on hand, respectively, for BIVIGAM, in excess of the distributors’ minimum requirements. These data are calculated using each distributor’s run rate for the previous month, demonstrating that ADMA’s immune globulin products are continuing to pull through the channel. Note: Due to year end calendar and fixed reporting schedules, January 5, 2026 is the closest available data for December 31, 2025.
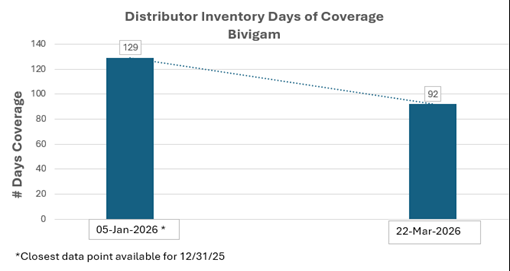
- As shown through data provided directly from ADMA’s distribution partners and direct customers, graphs entitled “Distributor Inventory Days of Coverage”, as of January 5, 2026 and March 22, 2026, illustrate the average of 128 and 90 days of inventory on hand, respectively, for ASCENIV, and 129 and 92 days of inventory on hand, respectively, for BIVIGAM, by ADMA’s distribution partners and direct customers, including the mutually agreed upon safety stock levels of ADMA’s immune globulin products. Note: Due to year end calendar and fixed reporting schedules, January 5, 2026 is the closest available data for December 31, 2025.
- The Company believes that these inventory levels are consistent with industry peers and are appropriately sized to ensure the cold-chain shipping, storage and handling of such products and to mitigate any potential supply chain or production disruptions.



- The Short Report misrepresents and conflates ASCENIV’s utility and competitive positioning in the IVIG market.
- ASCENIV is positioned as a late-line therapy for immune compromised individuals who are refractive to standard IG treatments, have comorbidities and suffer from chronic and persistent infections.
- As a late line therapy for patients who have failed standard IG, ASCENIV is in a different category and commands premium pricing. The Short Report’s citation of new launches of normal course immune globulin products as creating competitive pressure for ASCENIV utilization is grossly misguided.
- ADMA has received unqualified opinions in its audits for FY 2024 and FY2025.
- ADMA has filed its Annual Reports on Form 10-K for the years ended December 31, 2024 and 2025 with reports from a Big Four accounting firm that expressed unqualified opinions on the effectiveness of the Company’s internal control over financial reporting and an assessment that the Company’s consolidated financial statements are presented fairly, in all material respects, in conformity with U.S. generally accepted accounting principles (GAAP) for such periods.
- The Company’s prior accounting firm resigned in late 2024 in advance of being acquired by a private equity firm.
- There have never been any undisclosed related party transactions in violation of U.S. securities laws. No entity owned or controlled by Jerrold Grossman, Adam Grossman or the Grossman family has ever distributed, taken title to or otherwise possessed any of ADMA’s immune globulin or other products, whether for resale or otherwise. ADMA confirms that it has never sold any of its immune globulin products or other products to any entity controlled by the Grossman family or other related parties.
ADMA is committed to leading a new age of manufacturing, marketing, and commercializing specialty biologic products for the prevention and treatment of infectious diseases in the immune compromised and other patients at risk for certain infections, and creating long-term value for its stockholders. To receive accurate information about the Company, all investors are encouraged to review materials filed by ADMA with the U.S. Securities and Exchange Commission, which can also be accessed by visiting the Company’s website www.admabiologics.com.
About ASCENIV™
ASCENIV (immune globulin intravenous, human – slra 10% liquid) is a plasma-derived, polyclonal, intravenous immune globulin (IVIG). ASCENIV was approved by the United States Food and Drug Administration (FDA) in April 2019 and is indicated for the treatment of primary humoral immunodeficiency (PI), also known as primary immune deficiency disease (PIDD), in adults and adolescents (12 to 17 years of age). ASCENIV is manufactured using ADMA’s unique, patented plasma donor screening methodology and tailored plasma pooling design, which blends normal source plasma and respiratory syncytial virus (RSV) plasma obtained from donors tested using the Company’s proprietary microneutralization assay. ASCENIV contains naturally occurring polyclonal antibodies, which are proteins that are used by the body’s immune system to neutralize microbes such as bacteria and viruses that safeguard against infection and disease. ASCENIV is protected by numerous issued patents in the United States and internationally and a wide range of patent applications worldwide. Certain data and other information about ASCENIV can be found by visiting www.asceniv.com. Information about ADMA and its products can be found on the Company’s website at www.admabiologics.com.
Additional Important Safety Information About ASCENIV™
| WARNING: THROMBOSIS, RENAL DYSFUNCTION AND ACUTE RENAL FAILURE |
| Thrombosis may occur with immune globulin intravenous (IGIV) products, including ASCENIV. Risk factors may include: advanced age, prolonged immobilization, hypercoagulable conditions, history of venous or arterial thrombosis, use of estrogens, indwelling vascular catheters, hyperviscosity, and cardiovascular risk factors.
Renal dysfunction, acute renal failure, osmotic nephrosis, and death may occur with the administration of IGIV products in predisposed patients. Renal dysfunction and acute renal failure occur more commonly in patients receiving IGIV products containing sucrose. ASCENIV does not contain sucrose. For patients at risk of thrombosis, renal dysfunction or renal failure, administer ASCENIV at the minimum dose and infusion rate practicable. Ensure adequate hydration in patients before administration. Monitor for signs and symptoms of thrombosis and assess blood viscosity in patients at risk for hyperviscosity. |
ASCENIV™ Contraindications:
History of anaphylactic or severe systemic reactions to human immunoglobulin.
IgA deficient patients with antibodies to IgA and a history of hypersensitivity.
ASCENIV™ Warnings and Precautions:
IgA-deficient patients with antibodies against IgA are at greater risk of developing severe hypersensitivity and anaphylactic reactions. Have medications such as epinephrine available to treat any acute severe hypersensitivity reactions. [4, 5.1]
Thrombotic events have occurred in patients receiving IGIV treatments. Monitor patients with known risk factors for thrombotic events; consider baseline assessment of blood viscosity for patients at risk of hyperviscosity. [5.2, 5.4]
In patients at risk of developing acute renal failure. Monitor renal function, including blood urea nitrogen (BUN), serum creatinine, and urine output. [5.3, 5.9]
Hyperproteinemia, increased serum viscosity, and hyponatremia or pseudohyponatremia can occur in patients receiving IGIV treatment.
Aseptic meningitis syndrome (AMS) has been reported with IGIV treatments, especially with high doses or rapid infusion. [5.5]
Hemolytic anemia can develop subsequent to IGIV treatment. Monitor patients for hemolysis and hemolytic anemia. [5.6]
Monitor patients for pulmonary adverse reactions (Transfusion-related acute lung injury [TRALI]). If transfusion related acute lung injury is suspected, test the product and patient for antineutrophil antibodies. [5.7]
Because this product is made from human blood, it may carry a risk of transmitting infectious agents, e.g., viruses, and theoretically, the Creutzfeldt-Jakob disease (CJD) agent.
ASCENIV™ Adverse Reactions:
The most common adverse reactions to ASCENIV (≥5% of study subjects) were headache, sinusitis, diarrhea, gastroenteritis viral, nasopharyngitis, upper respiratory tract infection, bronchitis, and nausea.
To report SUSPECTED ADVERSE REACTIONS, contact ADMA Biologics at (800) 458-4244 or the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
About BIVIGAM®
BIVIGAM (immune globulin intravenous, human – 10% liquid) is a plasma-derived, polyclonal, intravenous immune globulin (IVIG). BIVIGAM was approved by the FDA in May 2019 and is indicated for the treatment of primary humoral immunodeficiency (PI), including, but not limited to the following group of genetic disorders: X-linked and congenital agammaglobulinemia, common variable immunodeficiency, Wiskott-Aldrich syndrome, and severe combined immunodeficiency. BIVIGAM contains a broad range of antibodies similar to those found in normal human plasma. These antibodies are directed against bacteria and viruses and help to protect PI patients against serious infections. BIVIGAM is a purified, sterile, ready-to-use preparation of concentrated human Immunoglobulin antibodies. Certain data and other information about BIVIGAM® or ADMA Biologics and its products can be found on the Company’s website at www.admabiologics.com.
Additional Important Safety Information About BIVIGAM®
| WARNING: THROMBOSIS, RENAL DYSFUNCTION AND ACUTE RENAL FAILURE |
| Thrombosis may occur with immune globulin intravenous (IGIV) products, including BIVIGAM. Risk factors may include: advanced age, prolonged immobilization, hypercoagulable conditions, a history of venous or arterial thrombosis, use of estrogens, indwelling vascular catheters, hyperviscosity and cardiovascular risk factors.
Renal dysfunction, acute renal failure, osmotic nephrosis, and death may occur with the administration of Immune Globulin Intravenous (Human) (IGIV) products in predisposed patients. Renal dysfunction and acute renal failure occur more commonly in patients receiving IGIV products containing sucrose. BIVIGAM does not contain sucrose. For patients at risk of thrombosis, renal dysfunction or renal failure, administer BIVIGAM at the minimum dose and infusion rate practicable. Ensure adequate hydration in patients before administration. Monitor for signs and symptoms of thrombosis and assess blood viscosity in patients at risk for hyperviscosity. |
BIVIGAM® Contraindications:
History of anaphylactic or severe systemic reactions to human immunoglobulin.
IgA deficient patients with antibodies to IgA and a history of hypersensitivity.
BIVIGAM® Warnings and Precautions:
Thrombotic events have occurred in patients receiving IGIV therapy. Monitor patients with known risk factors for thrombotic events; consider baseline assessment of blood viscosity for those at risk of hyperviscosity.
IgA deficient patients with antibodies against IgA are at greater risk of developing severe hypersensitivity and anaphylactic reactions. Have medications such as epinephrine available immediately to treat any acute severe hypersensitivity reactions.
Monitor renal function, including blood urea nitrogen (BUN), serum creatinine, and urine output in patients at risk of developing acute renal failure.
Hyperproteinemia, increased serum viscosity, and hyponatremia or pseudohyponatremia can occur in patients receiving IGIV therapy.
Aseptic meningitis syndrome (AMS) has been reported with IGIV treatments, especially with high doses or rapid infusion.
Hemolytic anemia can develop subsequent to treatment with IGIV products. Monitor patients for hemolysis and hemolytic anemia.
Monitor patients for pulmonary adverse reactions (Transfusion-related acute lung injury [TRALI]). If transfusion-related acute lung injury is suspected, test the product and patient for antineutrophil antibodies.
Because this product is made from human blood, it may carry a risk of transmitting infectious agents, e.g., viruses, and theoretically, the Creutzfeldt-Jakob disease (CJD) agent.
BIVIGAM® Adverse Reactions:
The most common adverse reactions to BIVIGAM (reported in ≥5% of clinical study subjects) were headache, fatigue, infusion site reaction, nausea, sinusitis, blood pressure increased, diarrhea, dizziness, and lethargy.
To report SUSPECTED ADVERSE REACTIONS, contact ADMA Biologics at (800) 458-4244 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
About ADMA Biologics, Inc. (ADMA)
ADMA Biologics is a U.S. based, end-to-end commercial biopharmaceutical company dedicated to manufacturing, marketing and developing specialty biologics for the treatment of immunodeficient patients at risk for infection and others at risk for certain infectious diseases. ADMA currently manufactures and markets three United States Food and Drug Administration (FDA)-approved plasma-derived biologics for the treatment of immune deficiencies and the prevention of certain infectious diseases: ASCENIV™ (immune globulin intravenous, human – slra 10% liquid) for the treatment of primary humoral immunodeficiency (PI); BIVIGAM® (immune globulin intravenous, human) for the treatment of PI; and NABI-HB® (hepatitis B immune globulin, human) to provide enhanced immunity against the hepatitis B virus. Additionally, ADMA is developing SG-001, a pre-clinical, investigative hyperimmune globulin targeting S. pneumonia. ADMA manufactures its immune globulin products and product candidates at its FDA-licensed plasma fractionation and purification facility located in Boca Raton, Florida. Through its ADMA BioCenters subsidiary, ADMA also operates as an FDA-approved source plasma collector in the U.S., which provides its blood plasma for the manufacture of its products and product candidates. ADMA’s mission is to manufacture, market and develop specialty plasma-derived, human immune globulins targeted to niche patient populations for the treatment and prevention of certain infectious diseases and management of immune compromised patient populations who suffer from an underlying immune deficiency, or who may be immune compromised for other medical reasons. ADMA holds numerous U.S. and foreign patents related to and encompassing various aspects of its products and product candidates. For more information, please visit www.admabiologics.com.
Cautionary Note Regarding Forward-Looking Statements
This press release contains “forward-looking statements” pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995, about ADMA Biologics, Inc. (“we,” “our” or the “Company”). Forward-looking statements include, without limitation, any statement that may predict, forecast, indicate, or imply future results, performance or achievements, and may contain such words as “confident,” “estimate,” “project,” “intend,” “forecast,” “target,” “anticipate,” “plan,” “planning,” “expect,” “believe,” “will,” “is likely,” “will likely,” “position us,” “positioned,” “support,” “should,” “could,” “would,” “may,” “potential,” “opportunity” or, in each case, their negative, or words or expressions of similar meaning. These forward-looking statements include, but are not limited to, statements about Short Report, including but not limited to the Company’s inventory levels and days on hand and product utilization. Actual events or results may differ materially from those described in this press release due to a number of important factors. Current and prospective security holders are cautioned that there also can be no assurance that the forward-looking statements included in this press release will prove to be accurate. Except to the extent required by applicable laws or rules, ADMA does not undertake any obligation to update any forward-looking statements or to announce revisions to any of the forward-looking statements. Forward-looking statements are subject to many risks, uncertainties and other factors that could cause our actual results, and the timing of certain events, to differ materially from any future results expressed or implied by the forward-looking statements, including, but not limited to, the risks and uncertainties described in our filings with the SEC, including our most recent reports on Form 10-K, 10-Q and 8-K, and any amendments thereto.
INVESTOR RELATIONS CONTACT:
Argot Partners | 212-600-1902 | [email protected]
MEDIA CONTACT:
Longacre Square Partners | [email protected]
Photos accompanying this announcement are available at
https://www.globenewswire.com/NewsRoom/AttachmentNg/fa2ef727-67b9-47b9-8903-3327560f7723
https://www.globenewswire.com/NewsRoom/AttachmentNg/1c4170ed-8dfa-46dc-87d2-4dc69bd58671
https://www.globenewswire.com/NewsRoom/AttachmentNg/cc3886f3-76e6-4df6-a915-1e5106dad222
https://www.globenewswire.com/NewsRoom/AttachmentNg/52920050-f97e-4b6f-aaec-4234d158f7ce
https://www.globenewswire.com/NewsRoom/AttachmentNg/c2e4dfc0-94d2-4de6-8ca0-8e790ba8a1ee

